Alliance
Alone we go faster, TOGETHER we go further !
The challenges are obviously technical and scientific, but they are definitely medico-economic. Therefore, developing cell-based therapies involve mastering technical, scientific, industrial manufacturing and regulatory constraints.
Mutualization of skills is essential in a field where the complexity of the issues is predominant.
This translates among other things into :
-
- One clear, detailed and unambiguous proposal
- A Technology Transfer driven by Communication, Co-working and Go/NoGo de-risking approach
- A constant flexibility in integrating unforeseen complementary studies, without necessarily contracting “Change of scope” Proposal
- A risk sharing to ensure successful target achievement
40+
Process diagnostics
1000+
QA documentations
10+
scale-up success
10+
cell types
20+
years of experience on ATMP development
30+
diagnostic of process
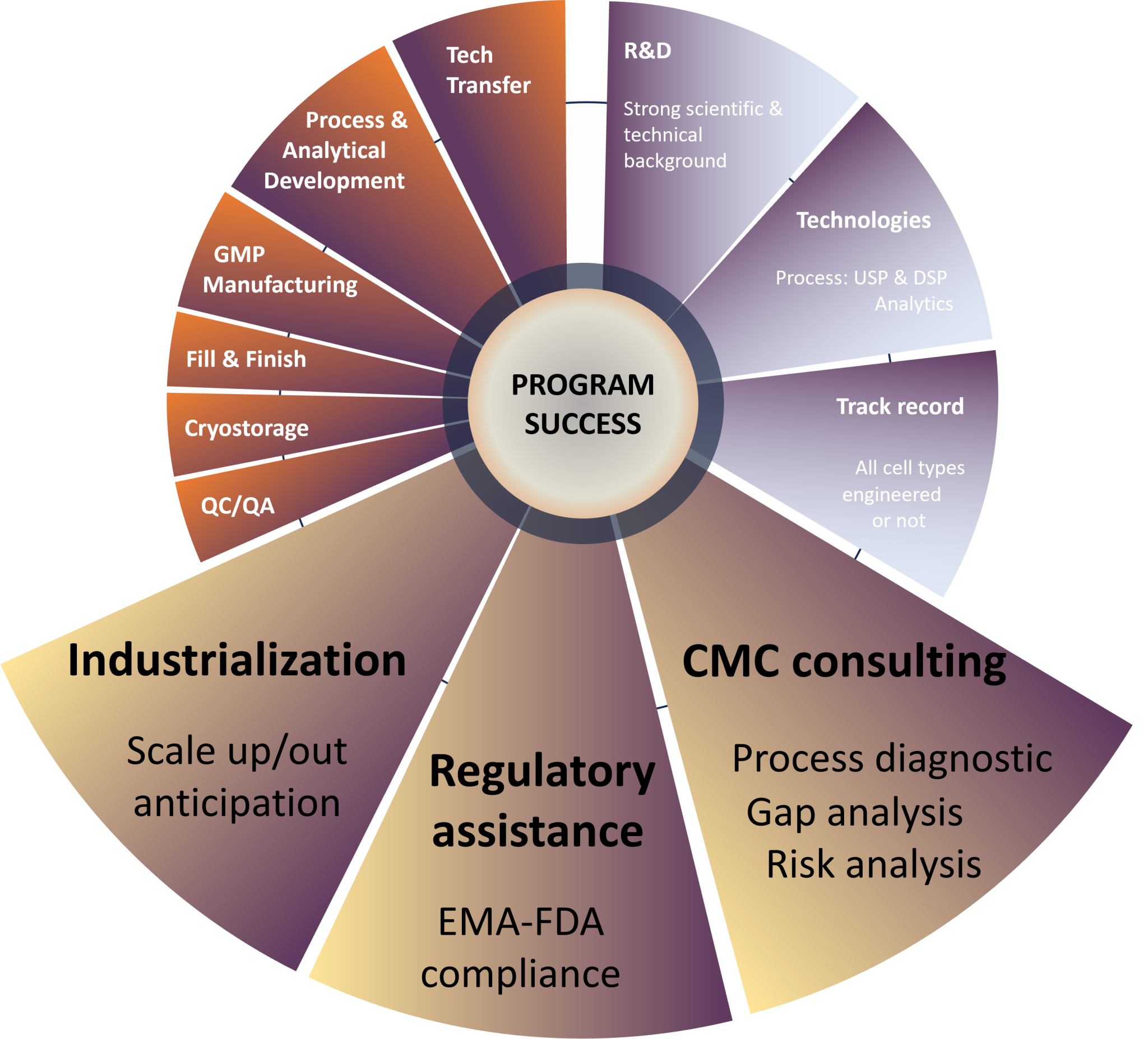
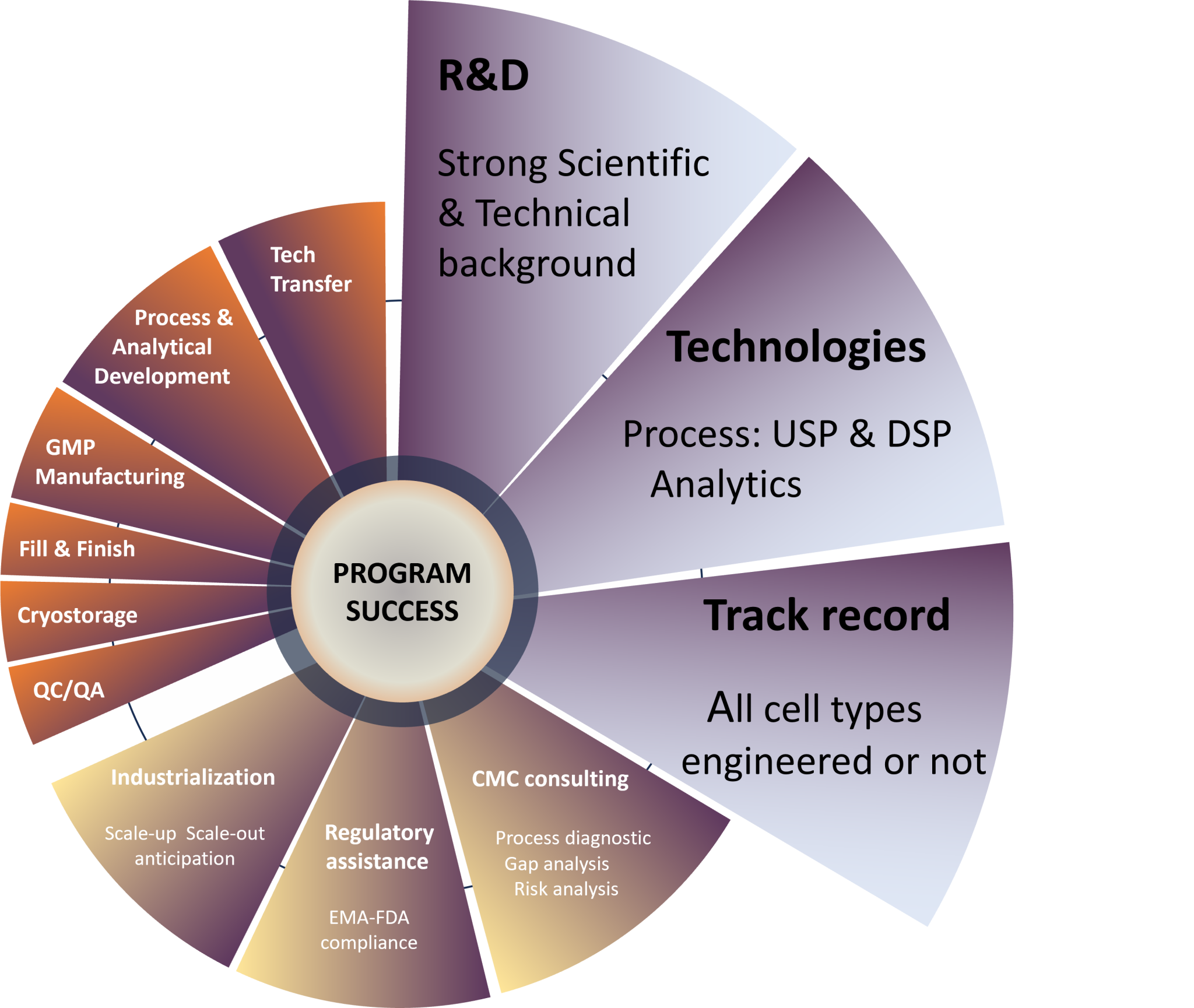
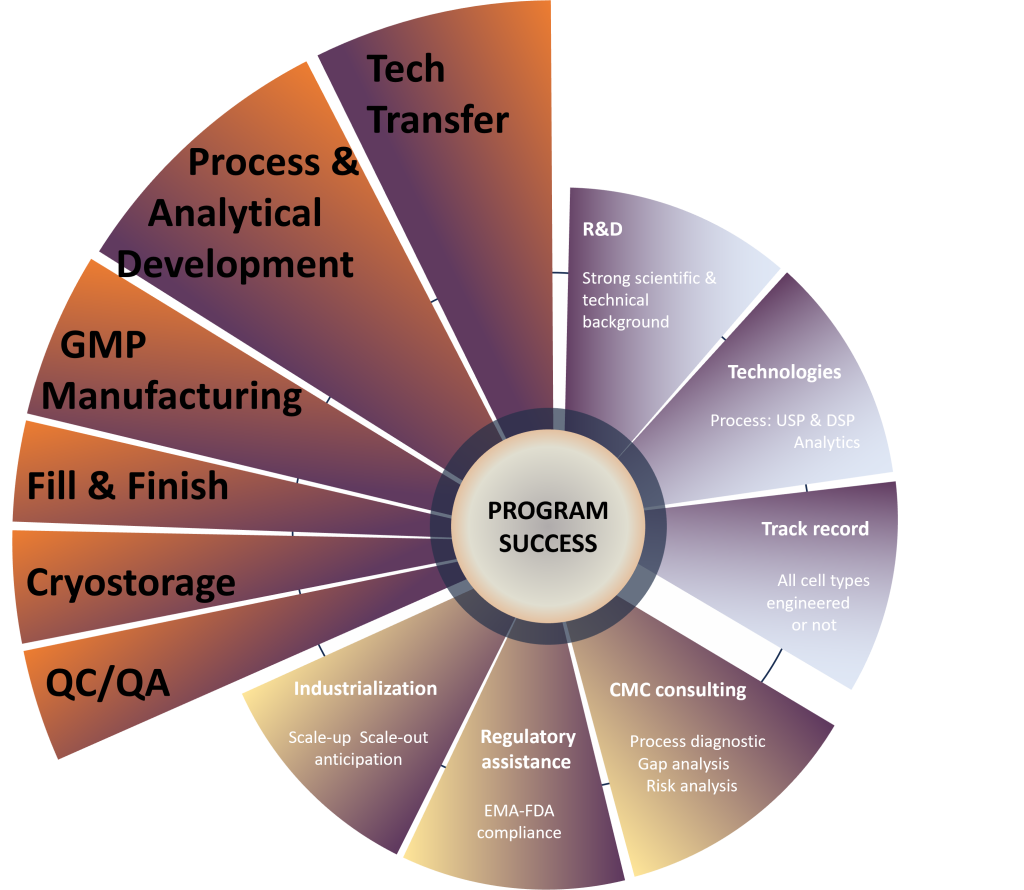
In pharmaceutical industry, the field of Cell Therapy is certainly the most complex in terms of both regulatory requirements and “Chemistry, Manufacturing, and Controls” (CMC).
Cell-based Products are living Drugs. That’s why it’s crucial to fully understand all aspects of the clinical development of ATMPs from start to finish.
At Cell-Easy, our multi-disciplinary teams focus on SCIENCE and TECHNOLOGIES to propose solutions that meet product specifications and regulatory standards.
Programs completed
Keep an eye on your Process
Because Process is nothing without Analytics, our R&D Team provide tailored analytical plan ranging into SAFETY, IDENTITY and POTENCY passports.
ATMP Expert
ART of
Manufacturing
ATMP is not conventional Drug Product
Unlike all other therapeutic products, cell-based products are considerably more complex to manufacture. In cell therapy, the PRODUCT is the PROCESS and the PROCESS is the PRODUCT !
It is essential to master the Art of aseptic manufacturing as ATMPs cannot undergo terminal sterilization.
50+
months of ATMP development
1 - 500+
Bag to Vials filling
100+
Technologies for Process & Analytics